When the proposed final drafts of USP <661.1> and <661.2> were published to USP39 NF34 in May, 2016, it was apparent that there were still many challenges to practical implementation of these new test requirements. As such, the effective for compliance was changed twice – first to May 1, 2020 and then further revised to December 1, 2025. Throughout this extensive and time consuming process, early adoption has been highly encouraged by the USP, regulatory bodies, and contract laboratories alike, including CS Analytical.
Although the general strategy and differences between the chapters is defined in more detail in the USP current revision, it is most straightforward to define <661.2> as being performed on the final package system – its method requiring that the final package be filled to its nominal volume for extraction. In comparison, <661.1> is intended to focus on materials of construction – this is done without respect to the volume of the final intended container. A known amount (mass) of plastic material is extracted in a constant volume of extraction media, most commonly water, with some variations on mass to volume ratios depending on the polymer. USP <661.1> also includes some more rigorous tests not found in <661.2>, such as extractable metals and plastic additive testing.
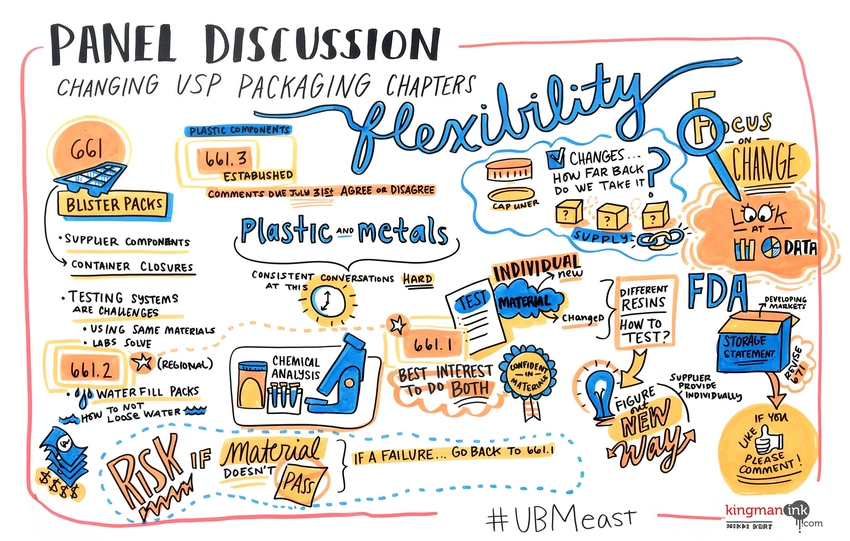 Amongst the challenges for practically implementing the new USP <661.1> and <661.2> was a defined approach for testing blister cards and their materials. This was a concept discussed in a June, 2016 conference session at EastPack Pharmaceutical and Medical Packaging 2016 in NYC . CS Analytical’s CSO Brandon Zurawlow participated on the panel with USP representatives and industry stakeholders, much of which became a Q&A for those in the audiences.
Amongst the challenges for practically implementing the new USP <661.1> and <661.2> was a defined approach for testing blister cards and their materials. This was a concept discussed in a June, 2016 conference session at EastPack Pharmaceutical and Medical Packaging 2016 in NYC . CS Analytical’s CSO Brandon Zurawlow participated on the panel with USP representatives and industry stakeholders, much of which became a Q&A for those in the audiences.
The primary issue regarding blister cards was filling sufficient cavities with water in order to yield enough solution for <661.2> testing. In order to perform the prescribed tests, at least 40mL of extraction solution is required. In the case of blisters, this could mean dozens or even hundreds of water-filled cavities would need to be prepared by the manufacturer for analysis.
Outside of the large number of samples required, logistically, preparing these samples has their own challenges. Due to the specialized, often custom process of forming and filling, and sealing blisters, this meant that many manufacturers would be required to shut down their line (or at least schedule time outside of production), manually fill sufficient cavities with water after forming, and send them for sealing. Many companies found this hard to get done from a production and quality perspective, while more still faced challenges with consistent filling using USP-grade water, or sealing due to water on critical surfaces.
While the option to create a “representative sample” with the same materials surface-area-to-volume ratio as the final blister design was and is allowed by <661.2>, designing tooling for a larger cavity that maintained these dimensional attributes remains both challenging and cost-ineffective.
The challenges in testing the final package system above ultimately led many companies to explore testing the forming material and lidding material separately by <661.1>. This too led to issues. However, this did not circumvent one glaring issue, that <661.1> alone was not intended to demonstrate the suitability of the final packaging system, which was to be established by <661.2>.
 Based upon feedback and comments as published in the 2019 PF the USP critically changed the scope of USP <661.2> from “this chapter applies specifically to plastic packaging systems. . .” to “this chapter applies specifically to plastic packaging components and systems. . . “. While this may be a small verbal change, the implications were wide. With the new proposed language, individual components could be tested directly to USP <661.2> specifications and demonstrated as “chemically suitable for its intended use”. However, this change actually muddied the waters and did nothing to solve the issue of specifically address the “blister card” issue.
Based upon feedback and comments as published in the 2019 PF the USP critically changed the scope of USP <661.2> from “this chapter applies specifically to plastic packaging systems. . .” to “this chapter applies specifically to plastic packaging components and systems. . . “. While this may be a small verbal change, the implications were wide. With the new proposed language, individual components could be tested directly to USP <661.2> specifications and demonstrated as “chemically suitable for its intended use”. However, this change actually muddied the waters and did nothing to solve the issue of specifically address the “blister card” issue.
As of 2022, it is the opinion of CS Analytical that the solution for testing blister cards is already present, but it is a matter of an experienced laboratory understanding the challenges, regulatory expectations, and the risk-based approach to container qualification testing that has been a dominant theme in USP chapter revisions over the last decade. With this, a solution can be developed and implemented.
As of October 2022, CS Analytical offers the ability to seal formed blisters in-house. Through collaboration with the client, custom tooling is acquired to seal blisters at lab-scale in our analytical laboratory, taking the burden off the client to acquire line time, expertly fill with USP-grade water, and introduce water to their filling and sealing operation. This is often more efficient from a time, cost, and regulatory perspective than designing tooling for a larger cavity that maintains the critical surface-area-to-volume ratio of the final configuration or justifying alternate approaches previously discussed.
With CSA’s solution to this challenge, a client sends us their blister cavity technical drawings, often required as part of the process for determining desiccant to be used for USP <671> testing, a related, but separate test set that evaluates permeation and light resistance of blister cards. CSA will determine, and the client will provide a sufficient formed blisters and lidding material to yield the total solution volume required for testing. At CSA, formed blister cavities are filled with USP-grade water using a micropipette, sealed, and placed into the extraction condition before being removed, cooled, and the resultant extract removed from each cavity and combined to perform all USP <661.2> tests.
CSA, with supporting comments from our clients, believes that this proposed approach provides a direct solution to the business, technical, and regulatory challenges companies can face when updating existing packaging data to USP <661.1> and <661.2>, or ensuring the success of new product-packaging coming to the market.

